
How many valence electrons sodium has, the number So the full electronĬonfiguration is 1S2, 2S2, 2P6, and 3S1. And you have one moreĮlectron to worry about. You to the third period or the third energy level. Write an electron configuration for sodium- IĪssume you already know how to do these- so you And so if we're talkingĪbout the main groups, the valence electronsĪre the electrons in the outermost shell or Ignore groups three through 12 for this video. So one, two, three, four,įive, six, seven, and eight. Through eight system for classifying groups. Valence electrons for elements in the main groups. Periodic table, let's see how to determine the But the octet rule is not always followed.Ĭlassified our elements into groups on the NOTE: The Pauli exclusion principle is one of the few laws that all of the elements always obey. You have to take all of the factors into consideration to predict how an element might behave. So, with chemistry, there are many competing factors that affect how elements behave, so you cannot just take a single rule and insist or suspect that every element must necessarily follow that rule. So, while the octet rule is a convenient rule of thumb, the reality of chemistry is much, much more complicated. There are many considerations as to what does or does not make an atom with a particular electron configuration reactive or nonreactive. There is no p subshell in shell number 1, so that is part of why the lightest elements do not follow the octet rule (there are other reasons). The octet rule follows from the energetic favorability of having both the s and p subshells either completely empty or completely full. Note: H₂ is stable, but it is quite reactive. Hydrogen does not follow the octet rule, so that is part of why H₂ is stable. So, the octet rule is not a strict law of science that is always followed. Other elements, such as S and P, sometimes do not follow the octet rule. 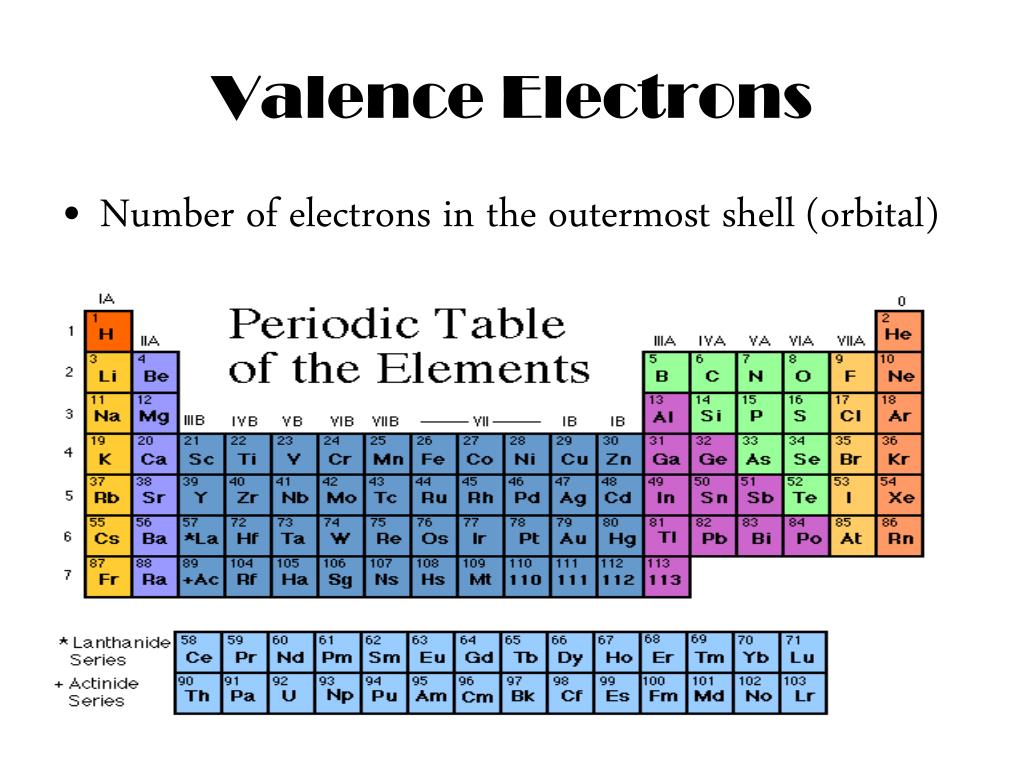
Boron usually does not follow the octet rule, though it sometimes does. H, He, Li, and Be never follow the Octet Rule. It is placed there because it is practically inert, like the other noble gases. We just place it there because it is unique and does not really fit into any group. Helium is not truly a member of Group 18 (or Group VIII in the older system).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
